Treatment & Management
When basal cell carcinoma (BCC) reaches an advanced stage, conventional treatments like surgery and radiotherapy may no longer be effective. In these cases, systemic treatments, including Hedgehog pathway inhibitors and immunotherapy, are essential.1
Targeted Therapy and Immunotherapy
Hedgehog Inhibitors (HHIs)
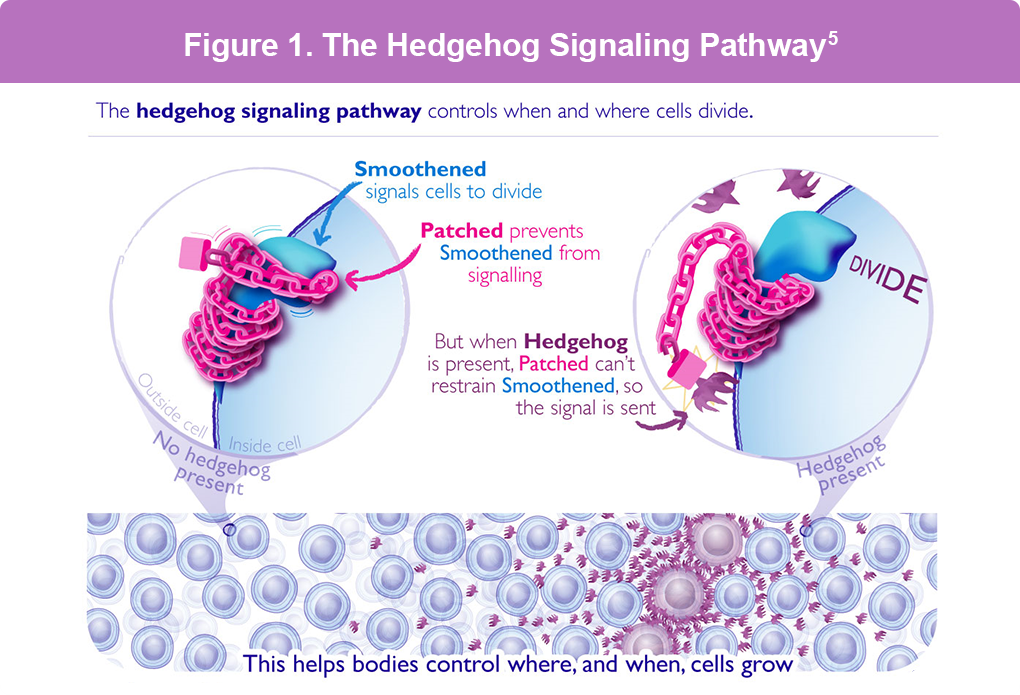
The Hedgehog pathway is a vital system that helps cells talk to each other and grow. When this system doesn’t work right, it can cause BCC to grow (Figure 1).2 To treat advanced BCC, doctors may use either vismodegib (Erivedge®) or sonidegib (Odomzo®), which help block this faulty cell communication and slow down cancer. These medicines are known as Hedgehog pathway inhibitors. Common side effects of these medications can include nausea, taste changes, muscle spasms, hair loss, weight loss, diarrhea, and fatigue.3,4
Figure 1. Normally, the smoothened molecule on a cell surface signals cells to divide/grow, and the patched molecule prevents them from growing by blocking smoothened from signaling. When Hedgehog is present, patched cannot restrain smoothened so cells continue to grow. In BCC, aberrant signaling of Hedgehog causes abnormal cell growth.5
Resistance to these drugs can develop, reducing their effectiveness.1 Serious side effects often lead to stopping treatment. To manage side effects and improve quality of life, drug holidays or alternative dosing schedules are recommended. If Hedgehog inhibitors are no longer effective or tolerated, immunotherapy with cemiplimab (Libtayo®) is recommended.1
Immunotherapy
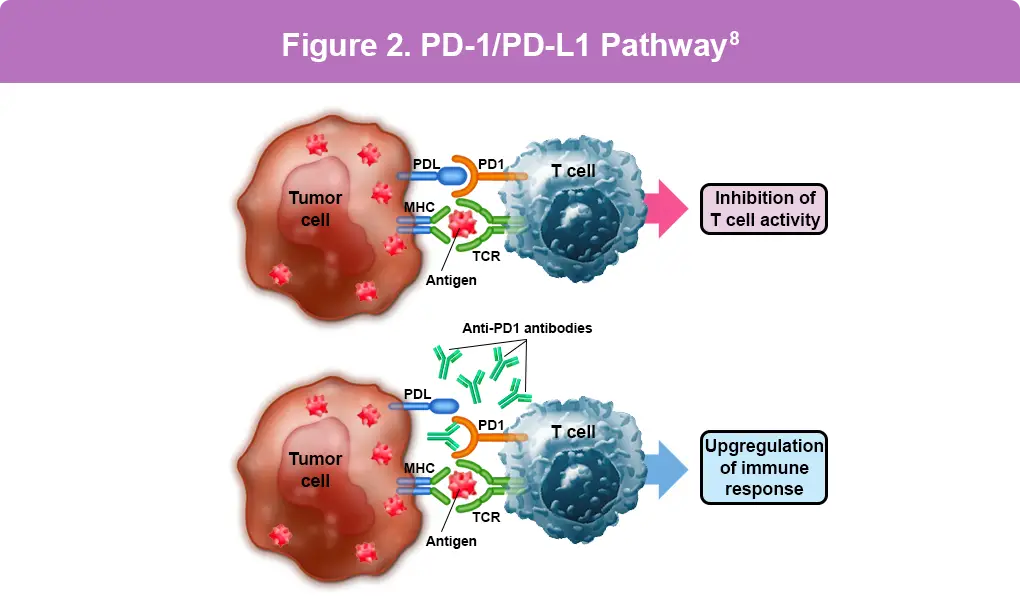
Immunotherapy drugs like cemiplimab (Libtayo®), pembrolizumab (Keytruda®), and nivolumab (Opdivo®) help the body’s immune system fight cancer. They work by targeting a part of immune cells called the PD-1 receptor. Normally, tumor cells use PD-L1 proteins to turn off immune cells, allowing the tumor cells to grow. These drugs block this process, allowing the immune cells to stay active and attack the cancer (Figure 2).2 Cemiplimab (Libtayo®), used for advanced BCC in people who do not respond to or cannot take Hedgehog inhibitors, has response rates between 22% and 31% for metastatic and locally advanced BCC. Common side effects of immunotherapy drugs include fatigue, musculoskeletal pain, diarrhea, rash, and itching.6,7
Figure 2. PD-L1 is found on tumor cells, while PD-1 is found on immune cells called T-cells. When PD-L1 on the tumor cell binds to PD-1 on the T-cell, it blocks essential cell survival signaling pathways, effectively inhibiting the cytotoxic T-cell. This inhibition deactivates the protective function of the T-cell, enabling the tumor cell to grow. Cemiplimab (Libtayo®) is an FDA-approved drug, while pembrolizumab (Keytruda®) and nivolumab (Opdivo®) have been and continue to be studied as potential options that work in the same way by blocking the interaction between PD-1 and PD-L1, allowing the immune cells to eliminate the tumor cells.8
Systemic treatments offer hope for managing advanced and metastatic BCC. Hedgehog pathway inhibitors are effective but have challenges like resistance and significant side effects. Immunotherapy provides an alternative for those who cannot tolerate or no longer respond to Hedgehog inhibitors.9
To achieve the best outcomes, treatment should follow established guidelines and involve a team of specialists to tailor the approach to each patient’s needs. Ongoing research aims to find new therapies with higher response rates and fewer side effects.
References
- National Comprehensive Cancer Network®. NCCN Clinical Practice Guidelines in Oncology. Basal Cell Skin Cancer. Version 2.2026. (https://www.nccn.org/professionals/physician_gls/pdf/nmsc.pdf).
- Bakshi A, Chaudhary SC, Rana M, Elmets CA, Athar M. Basal cell carcinoma pathogenesis and therapy involving hedgehog signaling and beyond. Mol Carcinog. 2017;56:2543-2557. doi:10.1002/mc.22690
- Sekulic A, Migden MR, Lewis K, et al. Pivotal ERIVANCE basal cell carcinoma (BCC) study: 12-month update of efficacy and safety of vismodegib in advanced BCC. J Am Acad Dermatol. 2015;72:1021-1026.e8. doi:10.1016/j.jaad.2015.03.021
- Migden MR, Guminski A, Gutzmer R, et al. Treatment with two different doses of sonidegib in patients with locally advanced or metastatic basal cell carcinoma (BOLT): A multicentre, randomised, double-blind phase 2 trial. Lancet Oncol. 2015;16:716-728. doi:10.1016/S1470-2045(15)70100-2
- Cancer Research UK. Cancer News. The story of vismodegib and skin cancer. June 7, 2012. (https://news.cancerresearchuk.org/2012/06/07/high-impact-science-hedgehogs-flies-and-skin-cancer-the-story-of-vismodegib/).
- Stratigos AJ, Sekulic A, Peris K, et al. Cemiplimab in locally advanced basal cell carcinoma after hedgehog inhibitor therapy: An open-label, multi-centre, single-arm, phase 2 trial. Lancet Oncol. 2021;22:848-857. doi:10.1016/S1470-2045(21)00126-1
- Lewis KD, Peris K, Sekulic A, et al. Final analysis of phase II results with cemiplimab in metastatic basal cell carcinoma after hedgehog pathway inhibitors. Ann Oncol. 2024;35(2):221-228. doi:10.1016/j.annonc.2023.10.123
- Angelousi A, Chatzellis E, Kaltsas G. New molecular, biological, and immunological agents inducing hypophysitis. Neuroendocrinology. 2018;106:89-100. doi:10.1159/000480086
- Work Group; Invited Reviewers, Kim JYS, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559. doi:10.1016/j.jaad.2017.10.006
All URLs accessed April 3, 2026